Publications
Independent Career
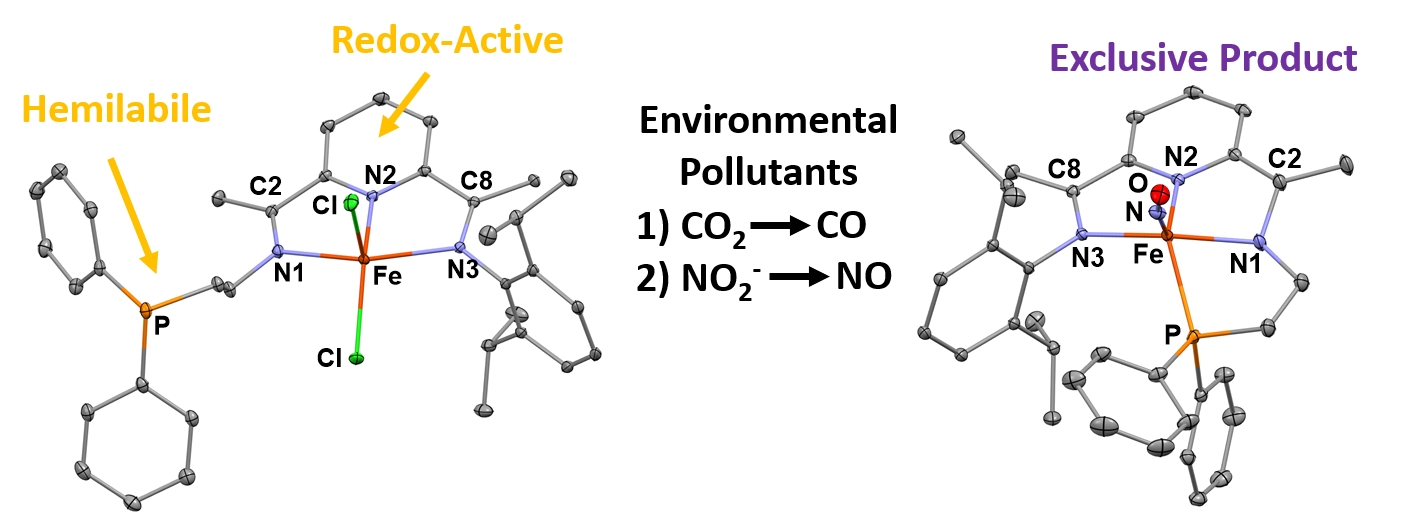
(31) Lewine, H. R.; Teigen, A.G.; Trausch, A. M.; Lindblom, K. M.; Seda, T.; Reinheimer, E.W.; Kowalczyk, T.; Gilbertson, J. D. Sequential Deoxygenation of CO2 and NO2- via Redox-Control of a Pyridinediimine Ligand with a Hemilabile Phosphine. Inorg. Chem. 2023, 62, 15173-15179 DOI:10.1021/acs.inorgchem.3c02323
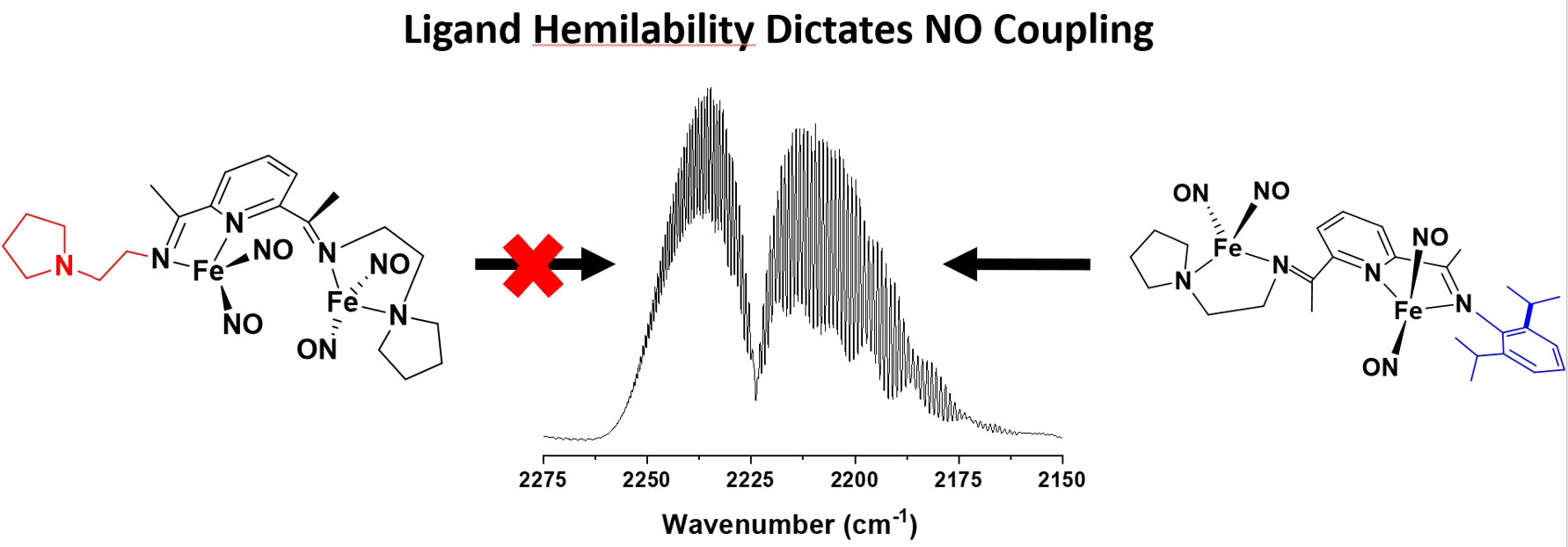
(30) Marks, W. R.; Reinheimer, E. W.; Seda, T.; Zakharov, L. N., Gilbertson, J. D. NO Coupling by Nonclassical Dinuclear Dinitrosyliron Complexes to Form N2O Dictated by Hemilability. Inorg. Chem. 2021, 60, 15901–15909 DOI:10.1021/acs.inorgchem.1c02285
(29) Marks, W. R.; Baumgardner, D.F.; Reinheimer, E. W.; Gilbertson J. D. Complete Denitrification of Nitrate and Nitrite to N2 Gas by Samarium(II) Iodide. Chem. Commun. 2020, 56, 11441-11444 DOI:10.1039/D0CC04115G
(28) Baumgardner, D. F.; Parks, W. E.; Gilbertson, J. D. Harnessing the Active Site Triad: Merging Hemilability, Proton Responsivity, and Ligand-Based Redox-Acitivty. Dalton Trans. 2020, 49, 960-965. DOI: 10.1039/C9DT04470A
(27) Cheung, P. M., Burns, K. T.; Kwon, Y. M.; Deshaye, M. Y.; Aguayo, K. J., Oswald, V. F.; Seda, T., Zakharov, L. N.; Kowalczyk, T.; Gilbertson, J. D. Hemilabile Proton Relays and Redox-Activity Lead to {FeNO}x and Significant Rate Enhancements in NO2- Reduction. J. Am. Chem. Soc. 2018, 140, 17040-17050. DOI: 10.1021/jacs.8b08520
(26) Burns, K. T.; Marks, W. R.; Cheung, P. M.; Seda, T.; Zakharov, L. N.; Gilbertson, J. D. Uncoupled Redox-Inactive Lewis Acids in the Secondary Coordination Sphere Entice Ligand-Based Nitrite Reduction. Inorg. Chem. 2018, 57, 9601-9610. DOI: 10.1021/acs.inorgchem.8b00032
(25) Delgado, M.; Gilbertson, J. D. Ligand-Based Reduction of Nitrate to Nitric Oxide Utilizing a Proton-Responsive Secondary Coordination Sphere. Chem. Commun. 2017, 53, 11249-11252. DOI: 10.1039/C7CC06541H
(24) Kwon, Y.; Delgado, M.; Zakharov, L.; Seda, T.; Gilbertson, J. D. Nitrite Reduction by a Pyridinediimine Complex with a Proton-Responsive Secondary Coordination Sphere. Chem. Commun. 2016, 52, 11016-11019. DOI: 10.1039/C6CC05962G
(23) Hartle, M. D.; Delgado, M.; Gilbertson, J. D.; Pluth, M. D. Stabilization of a Zn(II) Hydrosulfido Complex Utilizing a Hydrogen-Bond Accepting Ligand. Chem. Commun. 2016, 52, 7680-7682. DOI: 10.1039/C6CC01373B
(22) Cheung, P. M.; Berger, R. F.; Zakharov, L. N.; Gilbertson, J. D. Square Planar Cu(I) Stabilized by a Pyridinediimine Ligand. Chem. Commun. 2016,52, 4156-4159. DOI: 10.1039/C6CC00271D
(21) Delgado, M.; Ziegler, J. M.; Seda, T.; Zakharov, L. N.; Gilbertson, J. D. Pyridinediimine Iron Complexes with Pendant Redox-Inactive Metals Located in the Secondary Coordination Sphere. Inorg. Chem. 2016, 55, 555–557. DOI: 10.1021/acs.inorgchem.5b02544.
(20) Delgado, M.; Sommer, S. K.; Swanson, S. P.; Berger, R. F.; Seda, T.; Zakharov, L. N.; Gilbertson, J. D. Probing the Protonation State and the Redox-Active Sites of Pendant Base Iron(II) and Zinc(II) Pyridinediimine Complexes. Inorg. Chem. 2015, 54, 7239–7248. DOI: 10.1021/acs.inorgchem.5b00633
(19) Erickson, C. S.; Bradshaw, L. R.; McDowall, S. M.; Gilbertson, J. D.; Gamelin, D. R.; Patrick, D. L. Zero-Reabsorption Doped-Nanocrystal Luminescent Solar Concentrators. ACS Nano 2014, 8, 3461-3467. DOI: 10.1021/nn406360w
(18) Benjamin, W. E.; Veit, D. R.; Perkins, M. J.; Bain, E.; Scharnhorst, K.; McDowall, S.; Patrick, D. L.; Gilbertson, J. D. Sterically Engineered Perylene Dyes for High Efficiency Luminescent Solar Concentrators. Chem. Mater. 2014, 26, 1291-1293. Article selected as ACS Editor's Choice. DOI: 10.1021/cm403286v
(17) Thammavongsy, Z.; Breuhuas-Alvarez, A. G.; LeDoux, M.; Seda, T.; Zakharov, L. N.; Gilbertson, J. D. Pyridinediimine Iron Dicarbonyl Complexes with Pendant Lewis Bases and Lewis Acids Located in the Secondary Coordination Sphere. Eur. J. Inorg. Chem. 2013, 4008-4015. Invited Article for Special Cluster Issue on Small Molecule Activation. DOI: 10.1002/ejic.201300376
(16) Thammavongsy, Z.; Seda, T.; Zakharov, L. N.; Kaminsky, W.; Gilbertson, J. D. Ligand-Based Reduction of CO2 and Subsequent Release of CO on Iron(II) Inorg. Chem. 2012, 51, 9168–9170. DOI: 10.1021/ic3015404
(15) Kendall, A. J.; Zakharov, L. N.; Gilbertson, J. D. Synthesis and Stabilization of a Monomeric Iron(II) Hydroxo Complex via Intramolecular Hydrogen Bonding in the Secondary Coordination Sphere Inorg. Chem. 2010, 49, 8656–8658. DOI: 10.1021/ic101408e
Graduate and Postdoctoral Studies
(14) Chandler, B. D.; Long, C.; Gilbertson, J. D.; Pursell, C. J.; Vijayaraghavan, G.; Stevenson, K. J. Enhanced Oxygen Activation over Supported Bimetallic Au-Ni Catalysts. J. Phys. Chem. C, 2010, 114, 11498–11508.
(13) Yau, S. H.; Varnavski, O.; Gilbertson, J. D.; Chandler, B. D.; Ramakrishna, G.; Goodson, T. An Ultrafast Optical Study of Small Gold Monolayer Protected Clusters: A Closer Look at Emission. J. Phys. Chem. C, 2010, 114, 15979–15985.
(12) Long, C. G.; Gilbertson, J. D.; Vijayaraghavan, G.; Stevenson, K. J.; Chandler, B. D. Kinetic Characterization of Highly Active Supported Gold Catalysts from Monolayer Protected Clusters: An Experimental Michaelis-Menton Approach for Determining the Oxygen Binding Constant During CO Oxidation Catalysis. J. Am. Chem. Soc. 2008, 130, 10103-10115.
(11) Chandler, B. D.; Gilbertson, J. G. “Dendrimer-Encapsulated Nanoparticles in Catalysis” Invited book chapter in “Nanoparticles and Catalysis” p229-160, 2008 (Didier Astruc, Ed.), Wiley-VCH, Germany.
(10) Crump, C., Gilbertson, J. D.; Chandler, B. D. CO Oxidation and Toluene Hydrogenation by Pt/TiO2 Catalysts Prepared from Dendrimer Encapsulated Nanoparticle Precursors. Topics in Catalysis 2008, 49, 233-240.
(9) Gilbertson, J. D.; Vijayaraghavan, G.; Stevenson, K. J.; Chandler, B. D.; Air and Water Free Solid-Phase Synthesis of Soluble Nanoparticles with Anchored, Recyclable, Dendrimer Templates. Langmuir 2007, 23, 11239-11245.
(8) Gilbertson, J. D.; Szymczak, N. K.; Crossland, J. L.; Miller, W. K.; Lyon, D. K.; Foxman, B. M.; Davis, J.*; Tyler, D. R.; Coordination Chemistry of H2 and N2 in Aqueous Solution. Reactivity and Mechanistic Studies using trans-FeII(P2)2X2-type Complexes (P2 = a Chelating, Water-Solubilizing Phosphine). Inorg. Chem. 2007, 46, 1205-1214.
(7) Korkosz, R.; Gilbertson, J. D.; Prasifka, K.; Chandler, B. D. Dendrimer Templates for Supported Au Catalysts. Catalysis Today 2007, 122, 370-377.
(6) Chandler, B. D.; Gilbertson, J. D. “Dendrimer-Encapsulated Bimetallic Nanoparticles: Synthesis, Characterization, and Applications to Catalysis” Invited book chapter in “Dendrimer Catalysis” Topics in Organometallic Chemistry, vol. 21, p97-120, 2006 (Lutz Gade, Ed.), Springer, Heidelberg, Germany.
(5) Gilbertson, J. D.; Weakley, T. J. R.; Han, F.; Wolcott, J. J.; Tyler, D. R. Synthesis of ROMP Monomers Containing Metal-Metal Bonds. J. Inorg. Organomet. Polym. Mater. 2006, 15 (2005), 439-446.
(4) Gilbertson, J. D.; Szymczak, N. K.; Tyler, D. R. Reduction of N2 to Ammonia and Hydrazine Utilizing H2 as the Reductant. J. Am. Chem. Soc. 2005, 127, 10184-10185.
(3) Gilbertson, J. D.; Szymczak, N. K.; Tyler, D. R. H2 Activation in Aqueous Solution: Formation of trans-[Fe(DMeOPrPE)2H(H2)]+ via the Heterolysis of H2 in Water. Inorg. Chem. 2004, 43, 3341-3343.
(2) Miller, W. K.; Gilbertson, J. D.; Leiva-Paredes, C.; Bernatis, P. .; Weakley, T. J. R.; Lyon, D. K.; Tyler, D. R. Precursors to Water-Soluble Dinitrogen Carriers. Synthesis of Water-Soluble Complexes of Iron(II) Containing Water-Soluble Chelating Phosphine Ligands of the Type 1,2-Bis(bis(hydroxyalkyl)phosphino)ethane. Inorg. Chem. 2002, 41, 5453.
(1) Baxley, G T; Stiegman, A E; Nieckarz, G F; Weakley, T J; Tyler, D R; Gilbertson, J D. m-Bis(diphenylphosphino)methane-P:P'-octacarbonyldimanganese(Mn-Mn) and its toluene hemisolvate. Acta Cryst. 2001, C57, 1292.

