Faculty Research
Examples of ongoing projects include:
Synthetic Studies on the Archazolid Natural Products: The archazolids are a family of natural products that exhibit potent anti-cancer activity based on selective inhibition of the vacuolar-type ATPase (V-ATPase). We aim to better understand the archazolid-VATPase interaction through the development of efficient synthetic strategies and tractable V-ATPase assays.
diagrams of archazolid-VATPase interactions
References
-
King, B. R.; Swick, S. M.; Schaefer, S. L.; Welch, J. R.; Hunter, E. F.; O’Neil, G. W.* “Exploring a Ring-Closing Metathesis Approach to the Archazolid Macrocycle”, Synthesis 2014, 46, 2927-2936.
-
Swick, S. M.; Schaefer, S. L.; O’Neil, G. W.* “Synthesis of the C1-C17 fragment of the archazolids by complex cis-homodimer cross metathesis” Tetrahedron Lett. 2015, 56, 4039-4042.
Alkenone-Based Algal Biofuels: Long-chain alkenones are a unique class of lipids biosynthesized by several haptophyte algae. Alkenone structures are characterized by long hydrocarbon chains (C35 - C40) containing trans-double bonds separated by five methylenes and terminating in a methyl or ethyl ketone. We are investigating the potential for alkenones to serve as renewable sources of fuel and other valuable compounds.
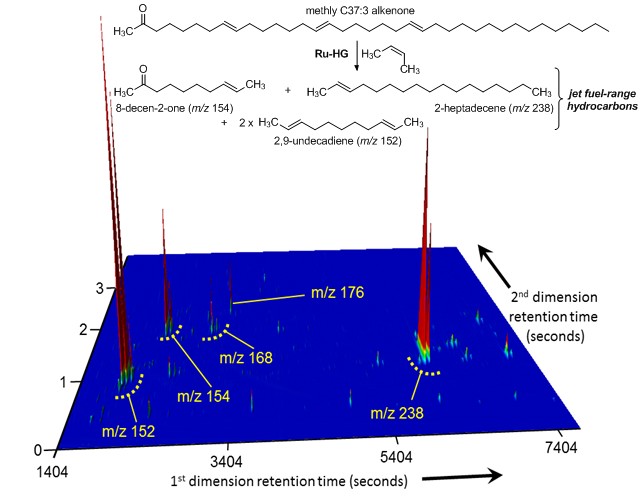
diagram of Long-chain alkenones
References
- O’Neil, G. W.*; Culler, A. R.; Williams, J. R.; Burlow, N. P.; Gilbert, G. J.; Carmichael, C. A.; Nelson, R. K.; Swarthout, R. F.; Reddy, C. M. “Production of Jet Fuel Range Hydrocarbons as a Coproduct of Algal Biodiesel by Butenolysis of Long-Chain Alkenones” Energy Fuels 2015, 29, 922-930.
-
O’Neil, G. W.*; Carmichael, C. A.; Goepfert, T. J.; Fulton, J. M.; Knothe, G.; Lau, C. P-L. Lindell, S. R.; Mohammady, N. G-E.; Van Mooy, B. A. S.; Reddy, C. M. “Beyond Fatty Acid Methyl Esters: Expanding the Renewable Carbon Profile with Alkenones from Isochrysis sp.” Energy Fuels, 2012, 26, 2434–2441.

